The role of radiation therapy in prostate cancer in New Zealand
Dr Ben Hindson, MBChB, FRANZCR, Christchurch
Dr Doug Iupati, MBChB, FRANZCR, Wellington
Prostate cancer remains New Zealand’s most common men’s cancer, with more than 3,500 diagnoses each year.1 Encouragingly, with advances in treatment and diagnostic tools in recent decades, prostate cancer has one of the highest survival rates of all cancers.
There is a wide variety of treatment options available to treat prostate cancer, particularly when detected early, including radiation therapy (external beam or brachytherapy) or surgery (radical prostatectomy). However, while these treatments have the same long-term survival outcomes, they may differ in side effect profile, quality of life impact, time and cost.
Radiation therapy continues to be underutilized in New Zealand, with the Royal Australian and New Zealand College of Radiologists (RANZCR) reporting that despite up to half of cancer patients being considered likely to benefit from radiation, less than a third will receive this type of treatment.2,3,4
This is also reflected in prostate cancer. In particular, brachytherapy can often be overlooked as a therapy option, due to a lack of knowledge about the treatment and its availability among healthcare professionals, despite its use in New Zealand for many years.
What is Brachytherapy?
Brachytherapy involves the placement of a radiation source in or near the cancer. It has been used by radiation oncologists for over 100 years (Figure 1.) and continues to be commonly used for treatment around the world today. There is a strong history of using brachytherapy in prostate cancer, perhaps related to the accessibility of the prostate gland.

Brachytherapy delivers a high radiation dose locally, with faster dose fall off peripherally. It also removes the problem of prostate movement during treatment, a common challenge faced during external beam radiation therapy (EBRT). Brachytherapy can be used either alone, or combined with EBRT to boost treatment dose.
In the treatment of prostate cancer, brachytherapy can be delivered one of two ways – High-Dose-Rate (HDR) or Low-Dose-Rate (LDR). HDR involves the temporary placement of a high activity (or high dose rate) radiation source, delivering a large dose of radiation to the prostate over 10-20 minutes. It is often combined with EBRT for higher risk cancers but also as monotherapy for intermediate risk cancers.
LDR brachytherapy is a well-established, minimally invasive treatment option for low or intermediate risk prostate cancer (T1-2N0M0; ISUP group 1 or 2 and PSA <20). LDR brachytherapy has been used in its modern form for over 30 years and continues to evolve. It involves permanently implanting 80-120 small radioactive Iodine125(I125) seeds, each the size of a grain of rice, into the prostate gland (Figure 2). These have low activity (low dose rate) and emit low energy radiation that is mostly absorbed by the prostate, with very little radiation emitted externally. The seeds remain in place, emitting very low doses of radiation over around a year. This approach destroys the tumour with minimum damage to the surrounding healthy tissue, and in a way which reduces disruption to ‘normal’ life.

LDR brachytherapy as a treatment option
Prostate cancer is a unique situation in which most treatment options, including surgery, EBRT and brachytherapy, offer equal cure rates.5 Each treatment option has specific impacts on the patient’s quality of life, including how long the course of treatment takes, recovery time, as well as the side effects associated with each treatment option.
LDR brachytherapy is associated with a lower risk of sexual and bladder problems and it is often able to be performed as a day-case procedure, with most men able to return to work the following week.6,7,8 This makes it an attractive option for men residing outside a major city or unable to take weeks off work, which can be a challenge faced with radical prostatectomy or other radiation therapy options.
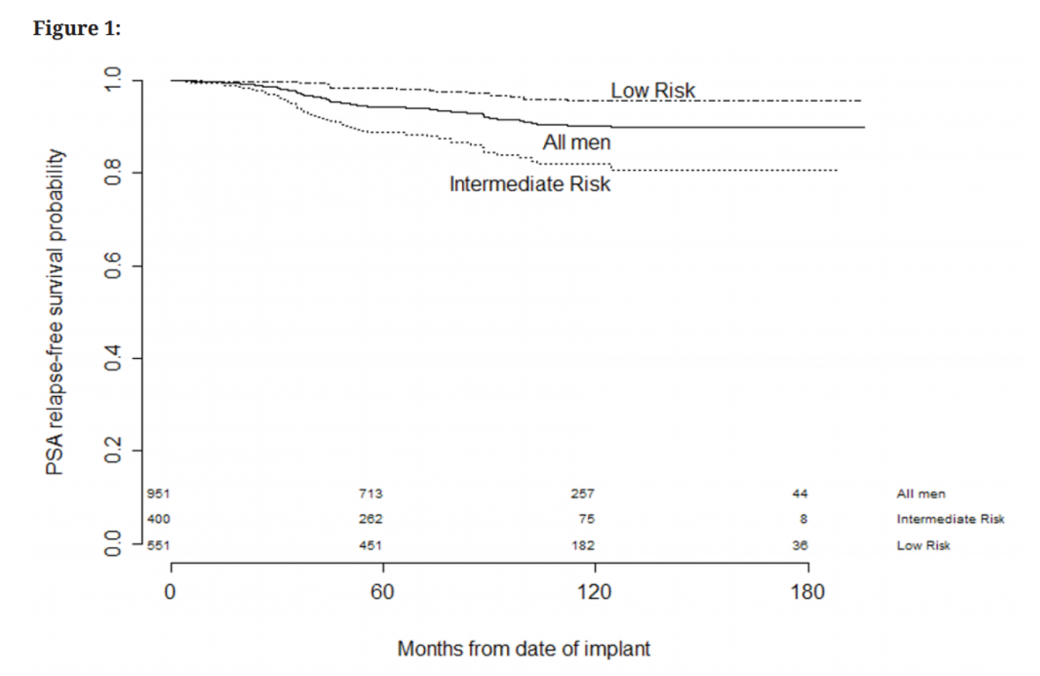
In both local and international published data on LDR brachytherapy, the 10-year PSA (prostate specific antigen) control is above 90% for all treated men.9,10 David Lamb and colleagues in Wellington published encouraging 5-year data demonstrating biochemical control of 95% for men with low-risk cancer and 82% for men with intermediate-risk cancer11 (figure 3), and several series have also shown the same durable disease control in younger men (<60 years).10,12
Erectile dysfunction (ED), urinary symptoms and bowel symptoms are side effects that need to be considered when treating prostate cancer, with ED often considered an important factor by patients. Perhaps the main advantage of LDR brachytherapy is the low risk of permanent side-effects.13 In the two months following LDR brachytherapy, men may experience temporary but significant urinary irritation and bother, however this can often be managed. Late urinary bother occurs in 10% of men, urethral strictures in 2-3%, with urinary incontinence not commonly seen. Bowel toxicity is also rare, with low occurrence of significant rectal bleeding or ulceration.10,11,13,14 Evidence suggests LDR brachytherapy offers the lowest risk to erectile function compared to many other prostate cancer treatments, with Ong et al reporting 60% of men preserved excellent erectile function at 5 years.15 In younger men, the erectile preservation rate was higher at 70%.
In New Zealand, LDR brachytherapy is a combined procedure performed by both a radiation oncologist and urologist. However, the procedure also requires a multidisciplinary team including radiation therapists, medical physicists, as well as the theatre staff. The procedure is performed under general anesthetic and usually takes about an hour, with patients able to return home on the same or following day.
Unfortunately, despite the well-established use of LDR brachytherapy in New Zealand and around the world, the treatment is currently only available through New Zealand’s private health system, and there is a need to improve public access to this option.
Making a treatment decision
The choice of prostate cancer treatment is a big decision for many men, and the amount of information on therapy options can be overwhelming. However, as prostate cancer is often slow growing, men and their families have an opportunity to explore all their treatments options.
Alongside urologists and radiation oncologists, GPs and practice nurses play a key role in the early detection of prostate cancer, the ongoing primary care of men being treated for the disease and importantly, empowering patients and their families to make informed treatment decisions and manage side effects.
Men diagnosed with prostate cancer should be encouraged to seek multidisciplinary consultation and speak with both a urologist and radiation oncologist before making a treatment decision. We hope to see increased awareness and improved access to all treatment options for New Zealand men diagnosed with prostate cancer.
Visit www.targetingcancer.co.nz for more information on radiation treatment options.
Literaturverzeichnis:
- Prostate Cancer Foundation New Zealand. Prostate Cancer. Available at: https://prostate.org.nz/prostate-cancer/.
- Barton M., Jacob S., Shafig J., Wong K., Thompson S., Hanna T., Delaney G. National & International Benchmarks set following study of delivery of Radiotherapy Services: ‘Review of Radiotherapy Optimal Utilisation Rates’. Collaboration for Cancer Outcomes Research and Evaluation (CCORE), Liverpool Hospital, Sydney, Australia, 2013: p6.
- Gabriel G, Barton M, Delaney G.Does travel distance affect radiotherapy utilisation in NSW and the ACT?Presentation at 2014 Innovations in Cancer Treatment and Care Conference.
- Royal Australasian College of Radiologists (RANZCR). Media statement: RANZCR welcomes progress in improving access to cancer care in New Zealand. 4 September 2019. Available at: https://www.ranzcr.com/documents-download/news-and-media/media-press-releases/4955-ranzcr-welcomes-progress-in-improving-access-to-cancer-care-in-nz-media-release/file.
- Hanady F.C., Donovan J.L., Lane J.A. et al. 10-Year Outcomes after Monitoring, Surgery, or Radiotherapy for Localized Prostate Cancer. N Engl J Med 2016;375:1415-24. DOI: 10.1056/NEJMoa1606220
- Grimm P et al. Comparative analysis of prostate-specific antigen free survival outcomes for patients with low, intermediate and high risk prostate cancer treatment by radical therapy. Results from the Prostate Cancer Results Study Group. BJU International 2012; 109, supplement 1: 22-29
- Malcolm, John B., et al. “Quality of life after open or robotic prostatectomy, cryoablation or brachytherapy for localized prostate cancer.” The Journal of urology 183.5 (2010): 1822-1829.
- Chen RC, Clark JA, Talcott JA. Individualizing quality-of-life outcomes reporting: how localized prostate cancer treatments affect patients with different levels of baseline urinary, bowel, and sexual function. J Clin Oncol. 2009;27(24):3916-3922.
- Routman D.M., Funk R.K., Stish B.J. Permanent prostate brachytherapy monotherapy with I-125 for low- and intermediate-risk prostate cancer: Outcomes in 974 patients. Brachytherapy 18 (2019) 1-7.
- Merrick G.S., Wallner K.E., Butler W.M. Brachytherapy in men aged <54 years with clinically localized prostate cancer. 2006 BJU INTERNATIONAL 98, 324–328.
- Lamb D.S., Greig L., Russell G. A prospective audit of the 10-year outcomes from low dose-rate brachytherapy for early stage prostate cancer. NZMJ 9 November 2018, Vol 131 No 1485.
- Langley S.E., Soares R., Uribe J. Long-term oncological outcomes and toxicity in 597 men aged ≤60 years at time of low-dose-rate brachytherapy for localised prostate cancer. BMJU Int 2018; 121: 38–45
- Keys M., M<iller S., Pickles T. Late Urinary Side Effects 10 Years After Low-Dose-Rate Prostate Brachytherapy: Population-Based Results From a Multiphysician Practice Treating With a Standardized Protocol and Uniform Dosimetric Goals. Int J Radiation Oncol Biol Phys, Vol. 90, No. 3, pp. 570e578, 2014
- Kishan AU, Kupelian PA. Late rectal toxicity after low-dose-rate brachytherapy: incidence, predictors, and management of side effects. Brachytherapy. 2015 Mar-Apr;14(2):148-59. doi: 10.1016/j.brachy.2014.11.005. Epub 2014 Dec 13. PMID: 25516492.
- Ong W.L., Hindson B.R., Beaufort C., Pharoah P., Millar J. Long-term erectile function following permanent seed brachytherapy treatment for localized prostate cancer. Radiotherapy and Oncology 112 (2014) 72–76
